Medicalinstruments for hyperthermia can be more patient-friendly and functional. Witha focus on different types of applicators and antennas, conventionalhyperthermia devices are reviewed by Manoshika Ramasamy, College of HumanEcology, Kansas State University, Manhattan and Minyoung Suh, Ph.D., College ofTextiles, North Carolina State University, Raleigh.
I. Introduction
Hyperthermiais a type of treatment that exposes cancer tissues to external heat to increaseits temperature to ~42 �C. The temperature between 41 �C and 44 �C is known to beimpactful, not to the normal cells, but to cancer cells. Hyperthermia is usedin combination with conventional therapies such as chemotherapy andradiotherapy. It helps drugs penetrate better into cancer cells in chemotherapyand makes them more sensitive to ionized radiation in radiotherapy. It hasclinical benefits to enable lower doses of drug and radiation use in cancertreatments. Ahmed & Zaidi (2013) reported a significantly reduced size of tumourcells when hyperthermia was combined with other treatments.
For thepast decades, several different devices for local hyperthermia have been developedand improved in order to achieve more promising clinical outcomes. Intraluminaland interstitial methods are used to treat tumours deep within the body, whereprobes or needles are struck inside the human body to approach to tumour cells.The energy source is then inserted into the probe to deliver heat to tumourcells directly. This method can minimise the heating damage to the normal tissuesnear the tumour cells, but its invasiveness makes it difficult to be acceptedto the wide variety of patients.
Non-invasiveapproaches are primarily developed to treat superficial tumours. Externaldevices are in contact with the skin of the tumorous region. Energy is appliedfrom the outside, targeting at the tumour to raise its temperature. Additionaldevice should be there to cool down the skin and normal tissues nearby, toprevent from getting burned.
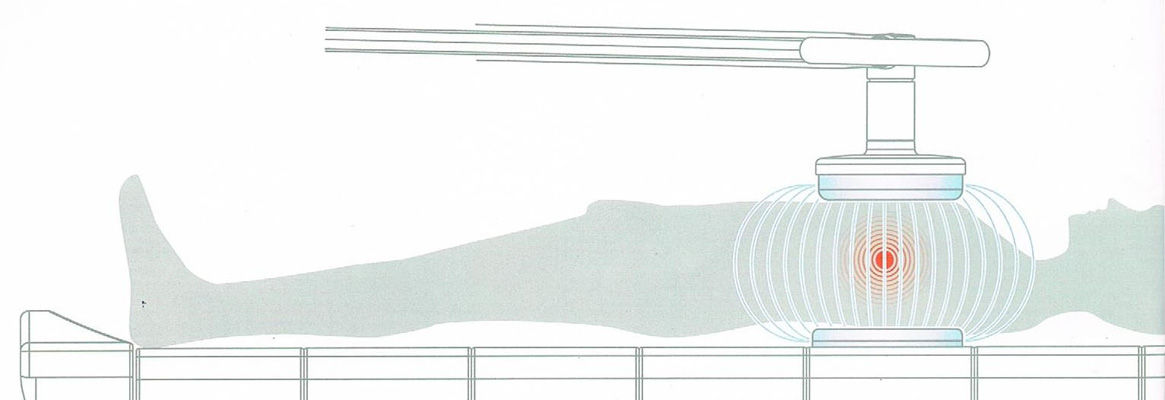
However, currentmedical instruments for non-invasive hyperthermia are not designed in apatient-friendly way. For example, a breast cancer patient must have heavymachinery placed on her body and place her breast into the cavity of the machineto expose tumorous regions to the heat source. She has to endure physicalhardship as well as heat stress while hyperthermia is in process. Thehyperthermia treatment lasts for at least one hour for a single treatment andis repeated on a regular basis depending on the advanced stages of tumour. Fromthe functional perspective, lacking flexibility and adjustability, currentdevices do not fit human body and it hinders uniform heating of irregularlyshaped body surfaces.
One of theapproaches for improvement is to take a wearable format. Related to user convenienceand treatment performance, there are a few benefits of the wearable device. First,the wearable device improves the physical environment of hyperthermia treatmentdramatically. A small and light wearable device can definitely give ease to thepatients that they do not have to endure the device throughout the treatment. Secondly,the device contoured along the human body shape enables more efficient andcontrolled heating simply because the distance between the antenna and cancercells becomes closer. In addition, it is possible to expect the synergic effectfrom small multiple antennas in different orientations.
The objective of this research is to show how the medical instruments for hyperthermia can be improved to be more patient-friendly and functional. With a focus on different types of applicators and antennas, conventional hyperthermia devices are reviewed. Important technical requirements for wearable hyperthermia devices are outlined and discussed accordingly. Based upon technical feasibility, it concludes that hyperthermia treatment can be administered in a wearable form by embedding heat-radiating elements into flexible substrates. The wearable hyperthermia device is considered as a possible solution to improve clinical environment in hyperthermia treatment.
Hyperthermia Device
Major components within a traditional hyperthermia device include antennas, a water bolus, and an applicator. An antenna is an element to radiate heat and the water bolus is necessary for better antenna coupling and cooling skin temperature down while the treatment continues. The applicator is the platform which houses antennas and water bolus. Typically, the applicator has a concave profile with an open cavity for electromagnetic waves. The antennas are functionally associated with the applicator and arrayed for transmitting heat energy at the selected amplitudes and relative phases into the cavity. Heat energy transmitted from the antennas passes through the water bolus to reach the tumour cell.
Applicator
Electromagnetic waves can be emitted through various shapes of waveguides such as a cuboid or a horn. A waveguide refers to a metal hollow pipe, which carries high frequency radio waves such as microwaves (Figure 1a). Since the heat is loaded in close connection to the area to be heated, this method does not cause excessive heating to non-cancerous areas, compared to other types of applicators. However, this applicator does not allow the patient to move while the treatment is in progress once the waveguide is positioned on the body. In addition, the shape of the aperture is not suitable for irregular body surfaces such as neck or axilla regions. According to Togni, Vrba, and Vannucci (2010), the heat is delivered in various tumour depths ranging from 6 to 15 mm by waveguide applicators.
Another type of applicator for local hyperthermia uses the water bolus itself as an applicator (Figure 1b). Antennas are mounted on the surface of water pouch. This type of applicator could be preferable due to a flexible and lightweight platform. The major advantages are potentials to incorporate multiple radiating elements and to treat wider areas compared to waveguide applicators.
A ring applicator (Figure 1c) is the movable unit mounted on a treatment bed. This is the way to heat relatively deep-located tumours, such as pelvis or abdomen. The adequate number of antennas is embedded inside of the ring around the patient. Desirable for regional hyperthermia, this applicator is feasible for body areas with irregular shapes. According to Wust et al. (2002), control of heat distribution can be significantly improved by increasing the number of antennas and optimising adjustment of phases and amplitudes.
Developed for whole body hyperthermia, a chamber applicator has a patient inside a moisture-saturated space thermally isolated from ambient environment (Figure 1d). It is possible to maintain a steady state of maximum temperatures of 42 �C for one hour with acceptable adverse effects. Since heat release through perspiration is blocked, the heating time can be shortened to 1 to 1.5 hours.
Antenna
An antenna is a unit to radiate electromagnetic waves within the hyperthermia device. Depending on the types of hyperthermia and its applicators, microwaves, radio frequency or infrared is used as an energy source. Most antennas in hyperthermia devices have worked at the frequencies of 27, 434, 915 or 2,450 MHz. Microwave energy is favourable to breast carcinoma which locates relatively less in depth, while radio frequency (RF) energy has the potential to achieve greater penetration due to relatively lower frequencies. There have been many different shapes of antennas developed for hyperthermia, such as monopole, dipole, loop, circular, rectangular, etc. The dimension of antenna or the elements with the antenna is designed according to the frequency in use.
Patch antennas employs a flat metal sheet as a radiating element and it is the most preferred antenna type in telecommunications due to its low profile and diverse polarisation. These antennas are constructed by different shapes of microstrip patch mounted on a dielectric surface. The patch has been fabricated by electroforming metal meshes or from commercially available planar conductive materials. The performance of the antenna varies according to the geometry of the patch and the thickness of the substrate.
Another important consideration related to the antenna performance is the presence of living cells in proximity. An antenna is known to experience significant changes in its performance when it is placed close to the human body. The body acted as a ground plane and influenced the radiation pattern. According to Alomainy et al. (2007), antenna detuning was different depending on antenna shapes. Small discrepancies between on-body and free space resonances had a great influence on the system reliability and efficiency. Other investigations dealt with electromagnetic interactions between antennas and biological tissues in different 3-dimensional orientations.
Water bolus
A water bolus has distilled water circulated to cool down the surface temperature to prevent skin burn. It also functions to obtain a better coupling between the antennas and the tissues. The water bolus is beneficial to reduce the size of antenna with the respect to resonant frequency. Therefore, property requirements of three major components, applicator, antenna, and water bolus, need to be well-identified and optimised with the consideration of mutual relations between these components.
Wearable Hyperthermia Device
Based upon the review of technical considerations related to hyperthermia applicators and antennas, a wearable device is proposed on which the small antennas are embedded into a conformable applicator. In previous research, it was found that textile antennas could function under the clothing environment associated with mechanical obstructions. However, unlike miniaturised wearable antennas widely used for telecommunication and entertainment purposes, the use of wearable antennas in medical fields has been limited to a sensing electrode to monitor human conditions and behaviours such as electrocardiogram or respiration. In this chapter, focusing on applicators and antennas, important characteristics are addressed to develop a wearable hyperthermia device.
Conformable applicator
Primarily, there should be a flexible applicator which is smaller in size and lighter in weight. Conforming to the body contour, the flexible applicator can reduce the distance between antennas and tumour tissues and this contributes to a more effective and controllable treatment. A pouch type applicator (Figure 1b) seems to have most potential for wearable applications, where the shell of water bolus is used as an applicator. Containing distilled water inside, a water bolus houses the antennas embedded on the surface.
Another important technical consideration for the applicator is thermal resistivity of the material. While the antenna is mounted on the substrate, it may have to get heat-treated and there might be a considerable amount of resistive heat generated on the antenna itself, which the substrate has to endure without being melted down. Kapton has been suggested for its superior thermal resistance. It is a polyimide film developed by DuPont that hardly melts (Dupont USA, 2014). However, Kapton is not favourable in terms of wearability due to its stiffness that does not allow it to conform along the curved surface easily.
In order to maximise user comfort, it would be ideal to use conventional textiles for underwear such as cotton jersey or stretch knits. Other alternatives include polymer films such as polyethylene terephthalate (PET) or polyvinyl chloride (PVC). Having less fuzzy exterior surfaces, synthetic polymers seem beneficial in terms of controlling conductive components embedded for antennas.
Printed antenna
The seamless integration of the antennas into conformable applicators is the next challenge. Conductive materials have been shifted or integrated into traditional textiles by weaving, knitting, stitching, couching, or printing. Weaving is a fabrication method to interlace two sets of conductive yarns in orthogonal directions, while knitting uses only one set of yarn. Through stitching, couching and printing, conductive materials can be added into the textile substrate during post-production processes.
Conductive printing has been used for many products and research in the field of wearable electronics and smart clothing. In contrast to patch antennas, a printed antenna can be obtained by depositing liquid conductive ink directly on the substrates. Printed on flexible polymeric substrate, the antenna adds less weight and bulk to the applicator. Compared to other methods, this method is favourable due to its capability to create delicate and complicated geometrical patterns such as thin spiral lines or multi-cornered shapes. With the ink-jet technology recently emerged, the printing process became quick and simple at fairly low manufacturing costs.
The design of antenna closely relates to the choice of applicator. The size, shape, number, and array of antennas should be decided considering the limited surface area of applicator. Multiple antennas can improve the treatment performance in a wearable device. The array of antennas needs to be carefully engineered in a way to avoid unexpected coupling or matching between them, which could be easily caused by the proximity of antennas. In-depth research would be necessary to establish proper proximity between antennas since the distance and orientation significantly change the antenna performance.
Treatment condition
Additional consideration related to the treatment performance is clinical conditions such as location, depth, and size of the tumour to be treated. For example, in case of ductal carcinoma which is the most common type of breast cancer, the tumour could be anywhere within the breast and its size is approximately 20 to 50 mm at Stage II. Therefore, the treatment for Stage II ductal carcinoma needs to be administered within a range of more than 20 mm in depth depending on the specific location of the tumour.
On the other hand, in recent trials to develop low-profile microwave antennas for hyperthermia, effective area of heat penetration was estimated to be 20 mm in depth with the treatment capability of tumour size at ~40 mm in diameter. Another experimental research showed that antennas could heat breast tissues up to 40 mm in depth when 18 small copper foil antennas were embedded into flexible polydimethylsiloxane (PDMS) substrate.
Hyperthermia seems to be technically feasible to be administered by the wearable device for some types of cancers. One single device would not be able to meet different levels of expectations in terms of treatment performance. If the tumours in specific depth or location are in consideration, the design of applicator and antennas could be optimised to yield best treatment outcomes.
This research explores the technical considerations for a wearable hyperthermia devices as an alternative technique of conventional hyperthermia treatment in clinics. Focusing on different types of applicators and antennas, possibility to incorporate them into the wearable platform was investigated. Critical design considerations and performance expectations are outlined in Table 1 and an expected design of the device is envisioned in Figure 2 for a breast cancer example.
|
Table 1. Technical considerations for wearable hyperthermia device | |||
|
|
Applicator (water bolus) |
Antenna |
Treatment condition |
|
Design decision |
Material Weight Bulk Thickness |
Fabrication method Frequency Shape and dimension Array of antennas |
Tumour size Location Depth |
|
Performance expectation |
Flexibility Thermal resistance Low cost |
Conductivity Return loss Radiation pattern |
Specific absorption rate (SAR) Temperature elevation Controllability |
|
|
|
Figure 2. Potential design example of wearable hyperthermia device for breast cancer patients |
Considering the technical feasibility related to hyperthermia devices, this study concludes that the wearable device is a possible alternative technique for hyperthermia treatments. Printing antennas on a conformable applicator could bring significant advantages to develop small, lightweight, cost-effective hyperthermia devices and this would significantly improve the quality of experience in clinics.
Acknowledgement
This work was supported by Johnson Cancer Centre and Kansas State University. The authors would like to thank Drs. Punit Prakash and Sergio Curto in Electrical and Computer Engineering, Kansas State University for their invaluable discussion and suggestions.
References:
1). Ahmed, K. and Zaidi, S. F. (2013, April). Treating cancer with heat: hyperthermia as promising strategy to enhance, Journal of Pakistan Medical Association, 63(4), 504-508.
2). Alomainy, A., Yang Hao, & Davenport, D. M. (2007). Parametric study of wearable antennas with varying distances from the body and different on-body positions. Paper presented at the Antennas and Propagation for Body-Centric Wireless Communications, April 24, 2007; London, UK.
3). Balanis, C. A. (2005). Antenna theory: Analysis and design. Hoboken, NJ: Wiley Inter science.
4). Curto, S., & Ammann, M. J. (2006). Electromagnetic interaction between resonant loop antenna and simulated biological tissue. Microwave and Optical Technology Letters, 48(12).
5). Dupont (2014). Summary of Properties for Kapton Polyimide Films, Dupont USA, Retrieved from http://www.dupont.com/content/dam/assets/products-and-services/membranes-films/assets/DEC-Kapton-summary-of-properties.pdf
6). Fenn, A. J. and Mon, J. (2004) Thermotherapy method for treatment and prevention of breast cancer and cancer in other organs, US Patent 6,690,976 B2.
7). Jacobsen, S., Rolfsnes, H. O., & Stauffer, P. R. (2005). Characteristics of microstrip muscle-loaded single-arm archimedean spiral antennas as investigated by FDTD numerical computations. IEEE Transactions on Biomedical Engineering, 52(2), 321-330.
8). Karaguzel, B., Merritt, C. R., Kang, T., Wilson, J. M., Nagle, H. T., Grant, E., and Pourdeyhimi, B. (2008) Utility of nonwovens in the production of integrated electrical circuits via printing conductive inks, Journal of the Textile Institute, 99(1), 37 - 45.
9). Korkmaz, E., Isik, O., and Nasoor, M. A. (2013). A Compact microstrip spiral antenna embedded in water bolus for hyperthermia applications. International Journal of Antennas and Propagation, 2013(2013), 1-6.
10). Moros, E. (2013). Physics of thermal therapy: Fundamentals and clinical applications. Boca Raton, FL: CRC, Taylor & Francis.
11). Paglione, R. W., Sterzer, F., Mendecki, J., Friedenthal, E., and Botstein, C. (1981). 27 MHz Waveguide Applicators for Localized Hyperthermia Treatment of Cancer. Paper Presented at IEEE International Conference on Microwave Symposium Digest, June 15-19, 1981; Los Angeles, CA, USA.
12). Patummakasorn, C., Tangwachirapan, S., & Thongsopa, C. (2008). Development of inductive regional heating by energy control at frequency 2.45 GHz, Paper Presented at 19th International Zurich Symposium on Electromagnetic Compatibility, May 19-23, 2008; Singapore.
13). Rychlik, O. & Vrba, J. (2008). Planar spiral applicator for local microwave thermotherapy, Paper Presented at 14th Conference on Microwave Techniques, April 23-24, 2008; Prague, Czech Republic.
14). Samulski, T. (2005) Non-invasive apparatus and method for providing RF energy-induced localized Hyperthermia, US Patent 6,904,323 B2.
15). Sharma, S., Bhushan, B., Gupta,S., Kaur, P. (2013). Performance comparison of micro strip antenna with different shape of the patch, International journal of u-and e- Service, Science and Technology, (6)3, 13-22.
16). Simon, H. & Zieve, D. (2012). Breast Cancer, University of Maryland, Medical Center, Retrieved on October 12, 2015 from https://umm.edu/health/medical/reports/articles/breast-cancer
17). Stauffer, P. R. and Maccarini, P. F. (2011). Evolution of antenna performance for applications in thermal medicine, Paper presented at 5th European Conference on Antennas and Propagation, April 11-15, 2011; Rome, Italy.
18). Suh, M., Carroll, K., Grant, E., & Oxenham, W. (2014). Investigation into Feasibility of Inductively Coupled Antenna for Smart Clothing, International Journal of Clothing Science and Technology, 26(1), 25-37.
19). Suh, M. (2010), E-Textiles for Wearability: Review of Integration Technologies, Textile World, Retrieved on October, 12, 2015 from http://www.textileworld.com/Articles/2010/April/Issue/Etextiles_Feature.html
20). Togni, P., Vrba, J., & Vannucci, L. (2010). Microwave applicator for hyperthermia treatment on in vivo melanoma model. Medical & Biological Engineering & Computing, 48(3), 285-292.
21). Topsakal, E., Asili, M., Chen, P., Demirci, U., & Younan, N. (2014). Flexible microwave antenna applicator for chemothermotherapy of the breast, Paper presented at 4th International Conference on Wireless Mobile Communication and Healthcare, November 3-5, 2014; Athens, Greece.
22). Wust, P., Hildebrandt, B., Sreenivasa, G., Rau, B., Gellermann, J., Riess, H., and Schlag, P. (2002). Hyperthermia in combined treatment of cancer. Lancet Oncology, 3(8), 487-497.
23). Zee, J. V., Holt, B. V., Rietveld, P. J., Helle, P. A., Wijnmaalen, A. J., Putten, W. L., and Rhoon, G. C. (1999). Re-irradiation combined with hyperthermia in recurrent breast cancer results in a worthwhile local palliation. British Journal of Cancer, 79(3-4), 483-490.
24). Zee, J. V. (2002). Heating the patient: a promising approach? Annals of Oncology, 13(8), 1173-1184.